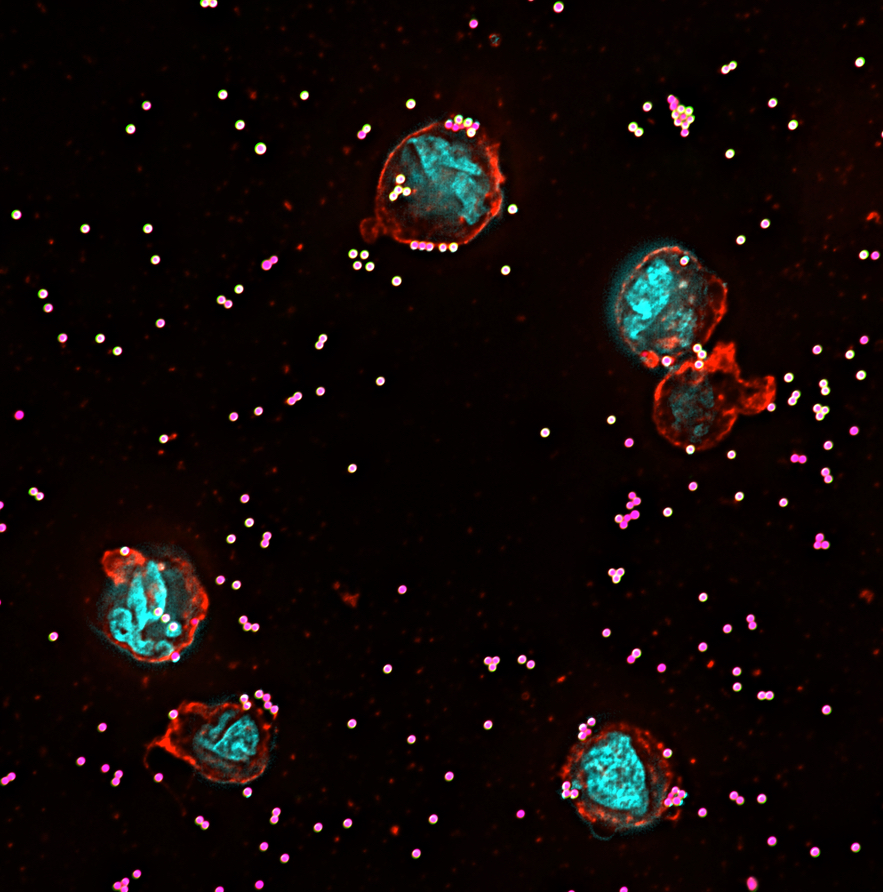
Research

The Nordenfelt Lab is interested in the interplay between human cells and their environment and the cell biology that controls the outcome of the interactions. This could be immune cells trying to find their way to invading bacteria and viruses or cancer cells migrating out of a tumor. Surface molecules, morphology, and biomechanical forces govern these interactions. Antibodies, the complement system, and the cellular receptors regulating signaling, such as Fc receptors and integrins, are natural components to study. As these are complex systems to study, the lab is continuously developing new technology to improve the range and quality of the research. The results from the research aim to improve our understanding as well as introduce new diagnostic and treatment options for infections and cancer.
Main areas of interest
Antibody function
The Nordenfelt Lab aims to increase our understanding of antibody function. We develop new human antibodies from patients, use a wide range of assays to assess their function, and subsequently improve them using antibody engineering. We are especially interested in the process of phagocytosis, including both Fc- and complement-mediated opsonization.
Key publications:
- Izadi, A., Karami, Y., Bratanis, E., Wrighton, S., Khakzad, H., Nyblom, M., Olofsson, B., Happonen, L., Tang, D., Sundwall, M., Godzwon, M., Chao, Y., Toledo, A. G., Schmidt, T., Ohlin, M., Nilges, M., Malmström, J., Bahnan, W., Shannon, O., Malmström, L. & Nordenfelt, P. The hinge-engineered IgG1-IgG3 hybrid subclass IgGh47 potently enhances Fc-mediated function of anti-streptococcal and SARS-CoV-2 antibodies.
Nature Communications. 2024. - Izadi, A., Hailu, A., Godzwon, M., Wrighton, S., Olofsson, B., Schmidt, T., Söderlund-Strand, A., Elder, E., Appelberg, S., Valsjö, M., Larsson, O., Wendel-Hansen, V., Ohlin, M., Bahnan, W., & Nordenfelt, P. Subclass-switched anti-spike IgG3 oligoclonal cocktails strongly enhance Fc-mediated opsonization.
PNAS. 2023. - Wrighton, S., Ahnlide, V.K., André, O., Bahnan, W., & Nordenfelt, P. Group A streptococci induce stronger M protein-fibronectin interaction when specific human antibodies are bound.
Frontiers in Microbiology. 2023. - Bahnan, W., Happonen, L., Khakzad, H., Ahnlide, V.K., Neergaard, T., Wrighton, S., André, O., Bratanis, E., Tang, D., Hellmark, T., Björck, L., Shannon, O., Malmström, L., Malmström, J., & Nordenfelt, P., 2022. A human monoclonal antibody bivalently binding two different epitopes in streptococcal M protein mediates immune function.
EMBO Molecular Medicine. 2022. - Bahnan, W., S. Wrighton, M. Sundwall, A. Bläckberg, O. Larsson, U. Höglund, H. Khakzad, M. Godzwon, M. Walle, E. Elder, A. S. Strand, L. Happonen, O. André, J. K. Ahnlide, T. Hellmark, V. Wendel-Hansen, R. PA. Wallin, J. Malmstöm, L. Malmström, M. Ohlin, M. Rasmussen, & Nordenfelt, P. Spike-Dependent Opsonization Indicates Both Dose-Dependent Inhibition of Phagocytosis and That Non-Neutralizing Antibodies Can Confer Protection to SARS-CoV-2.
Frontiers in Immunology. 2022. - de Neergaard, T., Bläckberg, A., Ivarsson, H., Thomasson, S., Kumra Ahnlide, V., Chowdhury, S., Khakzad, H., Bahnan, W., Malmström, J., Rasmussen, M., & Nordenfelt, P. Invasive streptococcal infection can lead to the generation of cross-strain opsonic antibodies.
Microbiology Spectrum. 2022. - de Neergaard T., Wrighton, S., Sundwall, M. & Nordenfelt, P. High-sensitivity assessment of phagocytosis by persistent association-based normalization.
Journal of Immunology. 2021. 206 (1) 214-224. - Kumra Ahnlide, V., de Neergaard, T., Sundwall, M., Ambjörnsson T., & Nordenfelt, P. A predictive model of antibody binding in the presence of IgG-interacting bacterial surface proteins.
Frontiers in Immunology. 2021. 12:661 - Nordenfelt, P., Waldemarson, S., Linder, A., Mörgelin, M., Karlsson, C., Malmström, J., & Björck, L. Antibody orientation at bacterial surfaces is related to invasive infection.
Journal of Experimental Medicine, 2012; 209(13), 2367–2381.
Cell migration and integrins
The Nordenfelt Lab aims to increase our understanding of how cell migration works in immunity and cancer. We use advanced microscopy, fluorescent sensor engineering, and image analysis to dissect molecular mechanisms of cell migration and integrins.
Key publications:
- André, O., Ahnlide, J.K., Norlin, N., Swaminathan, V., Nordenfelt, P. Data-driven microscopy allows for automated context-specific acquisition of high-fidelity image data. Cell Reports Methods. 2023.
- Nordenfelt, P., T. I. Moore, S. B. Mehta#, J. M. Kalappurakkal, V. Swaminathan, N. Koga, T. J. Lambert, D. Baker, J. C. Waters, R. Oldenbourg, T. Tani, S. Mayor, C. M. Waterman, and T. A. Springer. Direction of actin flow dictates integrin LFA-1 orientation during leukocyte migration.
Nature Communications. 2017. Dec; 8(1), pp. 2047. - Swaminathan, V., J. M. Kalappurakkal, S. B. Mehta, Nordenfelt P., T. I. Moore, K. Nobuyasu, D. A. Baker, R. Oldenbourg, T. Tani, S. Mayor, T. A. Springer, and Waterman, C.M. Actin retrograde flow actively aligns and orients ligand-engaged integrins in focal adhesions.
PNAS. 2017. 114 (40), pp. 10648–10653. - Nordenfelt, P., Elliott, H.L. and Springer T.A. Coordinated integrin activation by actin-mediated force during cell migration.
Nature Communications. 2016. Oct; 7(13119).
Microscopy and image analysis
The Nordenfelt Lab develops new technology for microscopy and image analysis. We use a programmatic approach to reduce the influence of human bias. This also increases both the throughput and quality of the microscopy. We also use fluorescence microscopy to measure and analyze molecular mechanisms.
Key publications:
- André, O., Ahnlide, J.K., Norlin, N., Swaminathan, V., Nordenfelt, P. Data-driven microscopy allows for automated context-specific acquisition of high-fidelity image data. Cell Reports Methods. 2023.
- Kumra Ahnlide, V., Kumra Ahnlide, J., Wrighton, S., Beech, J. P. & Nordenfelt, P. Nanoscale binding site localization by molecular distance estimation on native cell surfaces using topological image averaging.
eLife. 2022. - Mohanty, T., Sørensen, O.E. and Nordenfelt, P. NETQUANT: automated quantification of neutrophil extracellular traps.
Frontiers in Immunology. 2018. 8, p. 1999. - Nordenfelt, P., T. I. Moore, S. B. Mehta#, J. M. Kalappurakkal, V. Swaminathan, N. Koga, T. J. Lambert, D. Baker, J. C. Waters, R. Oldenbourg, T. Tani, S. Mayor, C. M. Waterman, and T. A. Springer. Direction of actin flow dictates integrin LFA-1 orientation during leukocyte migration.
Nature Communications. 2017. Dec; 8(1), pp. 2047. - Nordenfelt, P., Elliott, H.L. and Springer T.A. Coordinated integrin activation by actin-mediated force during cell migration.
Nature Communications. 2016. Oct; 7(13119).